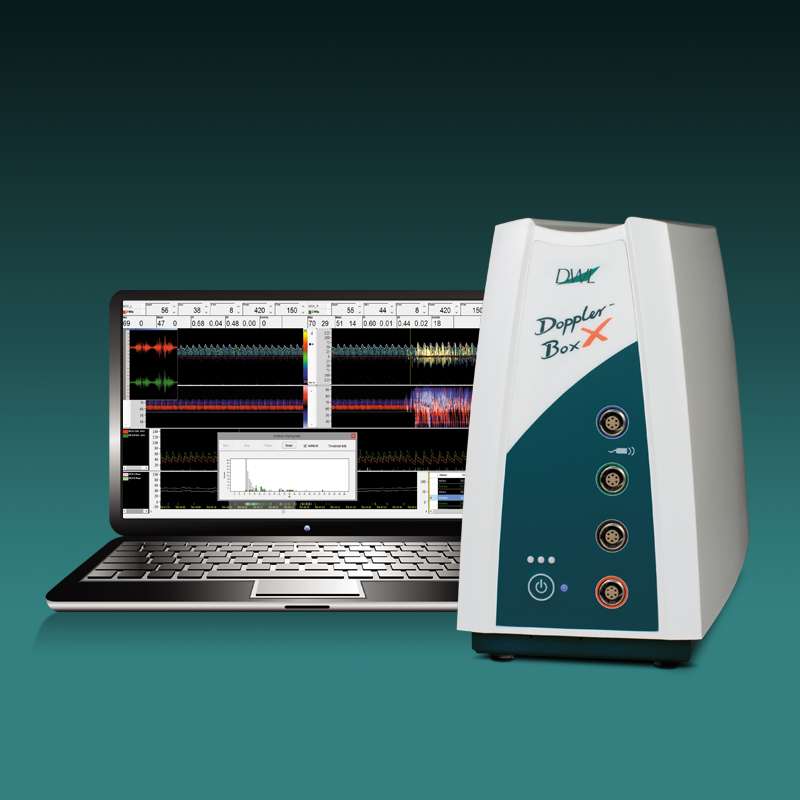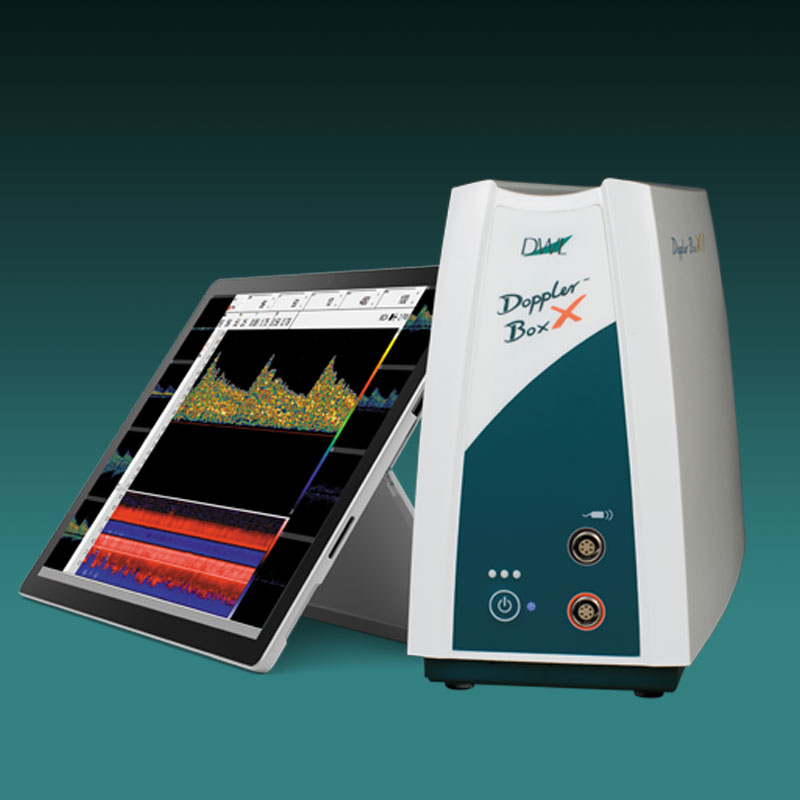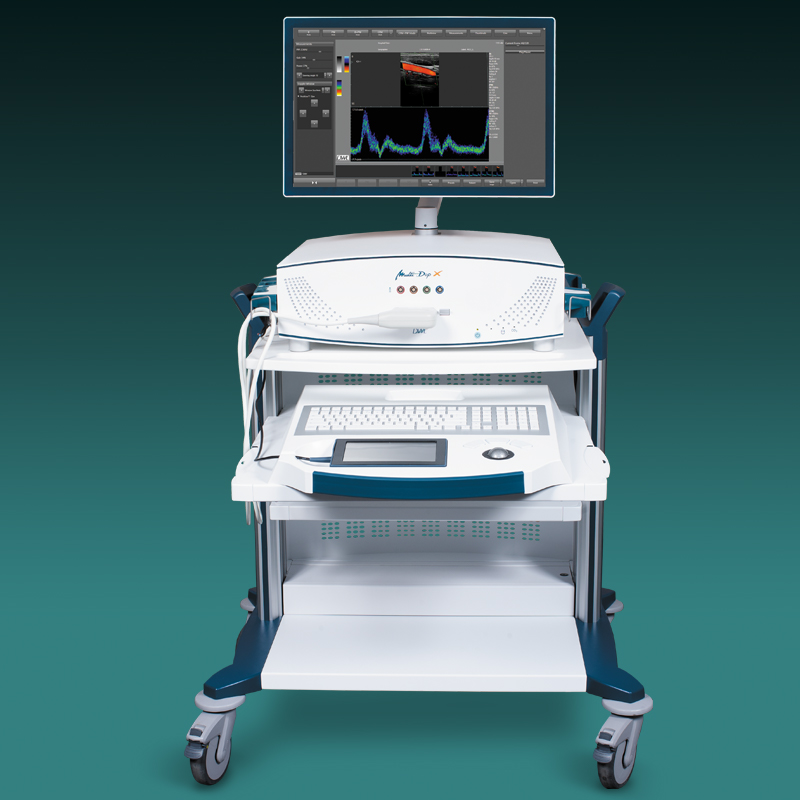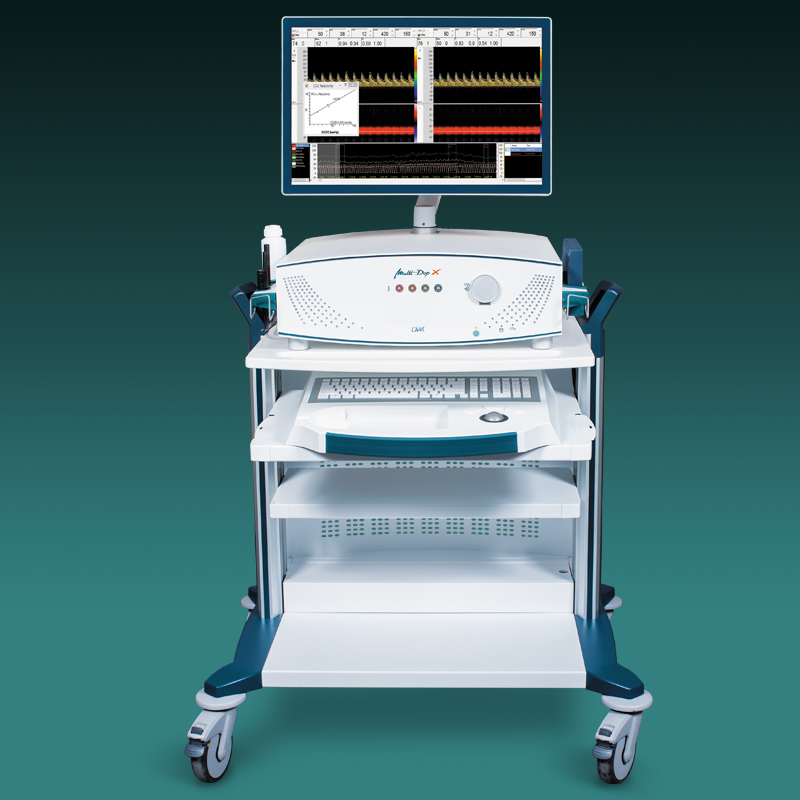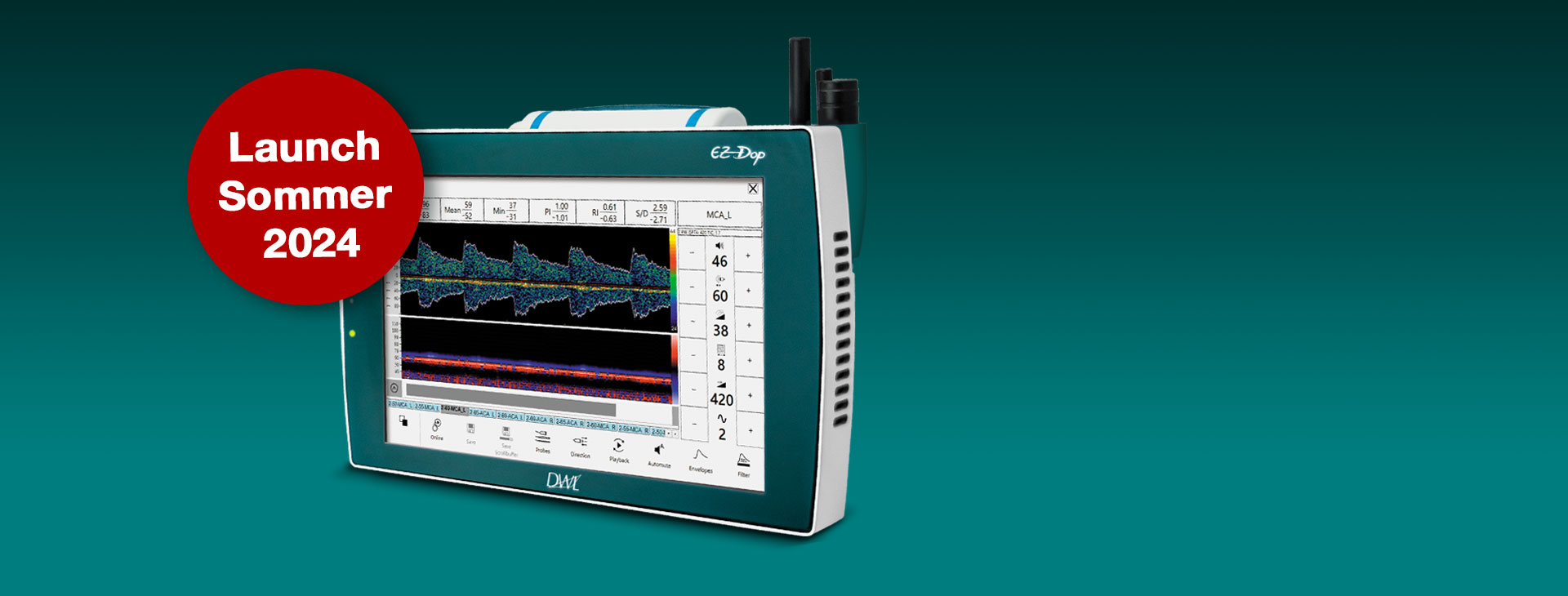
Der neue EZ-Dop
Das patientennahe Sofortdiagnostik-System für Untersuchungen direkt am Ort der Patientenversorgung.
mehr
Neuro-Monitoring-Analyse
TCD-Waveform Analyse Software mit Z-Score Auswertungsmodul in der kardiovaskulären Physiologie.
mehr
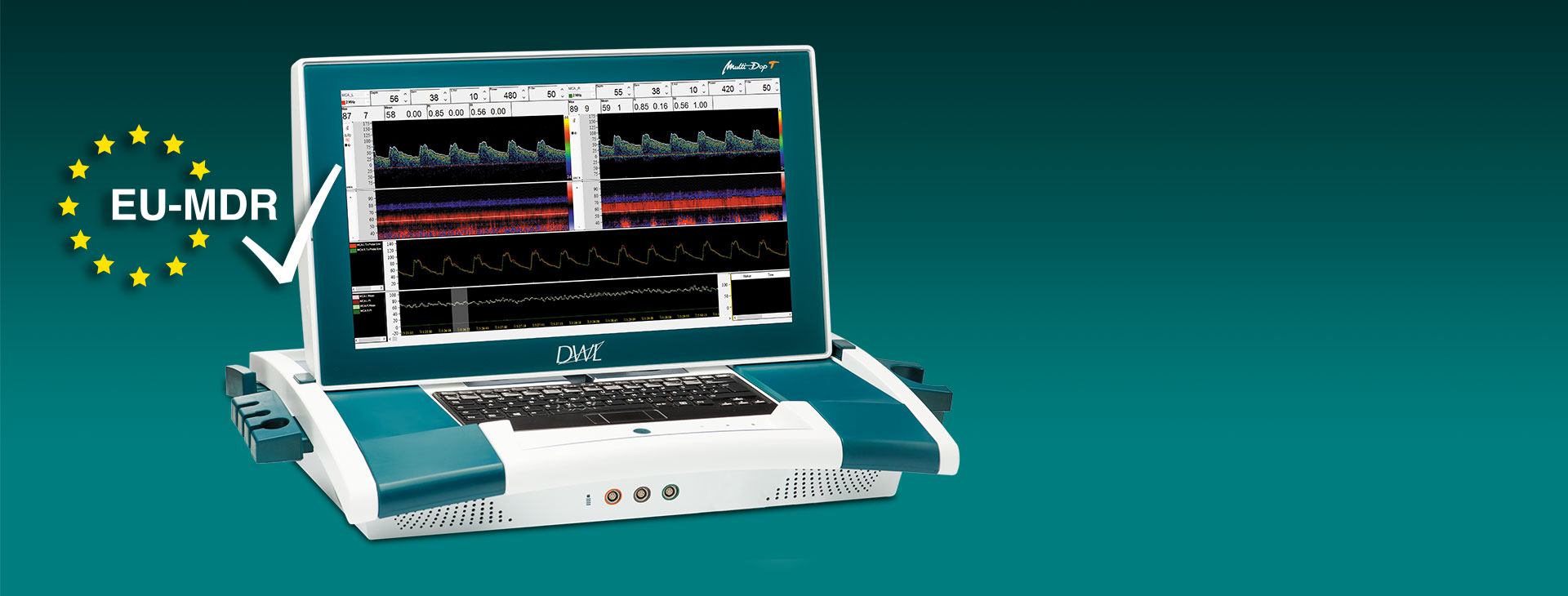
Vertrauen in Qualität und Sicherheit:
Das erste Modell der DWL-Produktpalette, der DWL Multi-Dop T, hat die Zertifizierung nach der MDR (EU) 2017/745 erhalten.
mehr
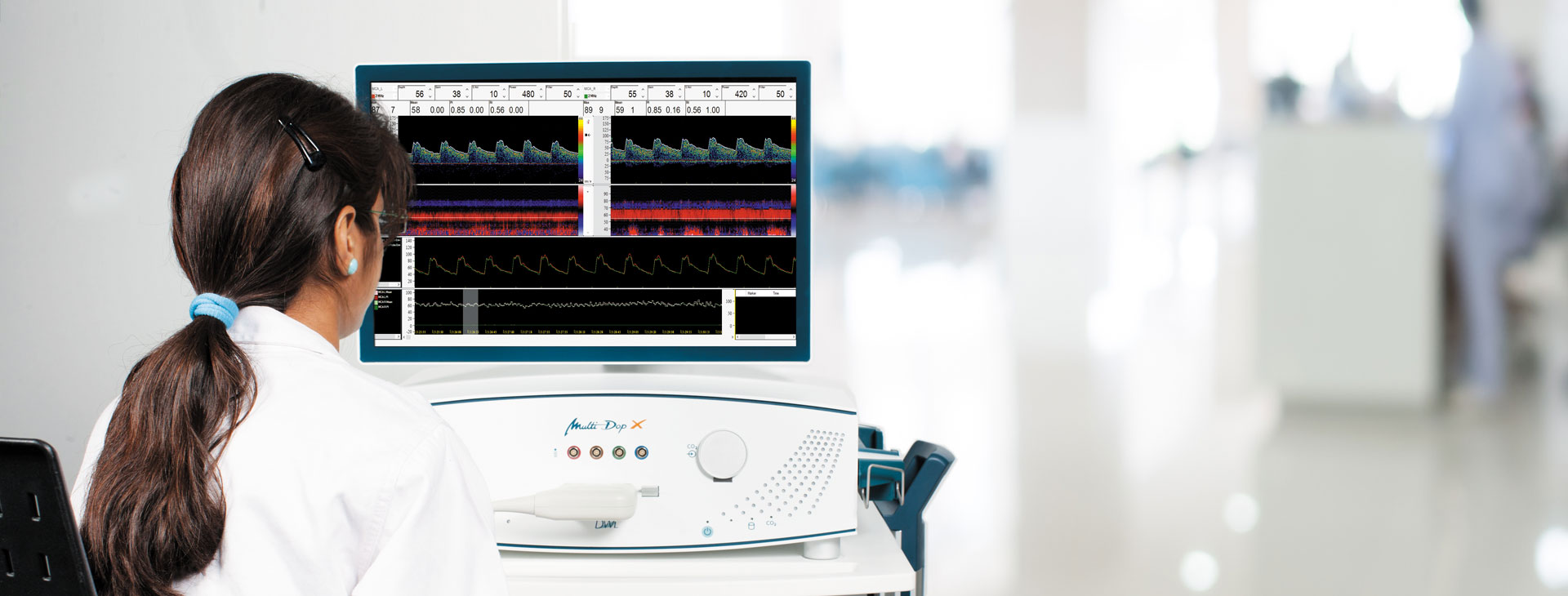
Leistung nach Maß
Vom portablen Basissystem bis hin zur Komplettlösung mit umfassenden Leistungsspektrum – DWL bietet Leistung nach Maß.
mehr

Kompetenz im OP
Flexibel, anwenderfreundlich, präzise und aussagekräftig – die DWL Dopplersonographie hilft Leben retten.
mehr

International vor Ort
Durch unsere Unterstützung bei Fortbildungsveranstaltungen und der Teilnahme an einschlägigen Fachkongressen sind wir immer in Ihrer Nähe.
Mehr
Compumedics Germany / DWL
Entwicklung und Herstellung der Transkraniellen Dopplersonographie TCD – unsere Kernkompetenz.
DWL – Mit einer klaren Vision und mit einem hohen Anspruch wurde unser Unternehmen gegründet. Von den Anfängen im Jahre 1991 an lag unser Fokus auf der Weiterentwicklung und Optimierung der transkraniellen Dopplersonographie (TCD). Mit jeder neuen Systemgeneration wird die Dopplersonographie flexibler und anwenderfreundlicher, ihre Reports für die Befundung präziser und aussagekräftiger. Insbesondere durch die volldigitale Dopplersonographie mit M-Mode kommen unsere Systeme heute weltweit in einer Vielzahl oft lebensrettender Anwendungen zum Einsatz.
The World of TCD Clinical Applications
Hier finden Sie aktuelle Artikel, basierend auf klinischer Forschung und Veröffentlichung zu TCD in der Anwendungen, Diagnostik und bei Behandlungsmethoden.
-
TCD Clinical Indicationsam 7. März 2024
TCD provides noninvasive monitoring in diverse clinical situations, offering real-time physiological data without increasing patient risks or costs. Der Beitrag TCD Clinical Indications erschien […]
-
SARS-CoV-2 – update 2023am 2. November 2023
If the coronavirus causes vascular inflammation (vasculitis), the inflammatory reaction leads to vascular narrowing with relevant cerebral blood flow velocities (CBFV) changes. An […]
-
Doppler and Duplexam 10. Juli 2023
Transcranial Doppler Sonography (TCD) and Transcranial Color Duplex Sonography (TCCD) are complementary diagnostic methods. The diagnosis of stenoses can be made with conventional TCD with the help […]
Die Vielfalt an klinischen Anwendungen der Dopplersonographie
Ob in der Neurologie, der Neuro-, Herz- und Gefäßchirurgie, in der Anästhesie, der Notfall- und Intensivmedizin sowie der HNO- und MKG-Chirurgie – das Anwendungsspektrum der DWL Dopplersysteme ist heute breiter als je zuvor.
Schlaganfall Management
Anästhesie
Sichelzellanämie Diagnose
Hirntod Diagnose
PFO Diagnose
Vasospasmen Diagnose
DWL Software für
Ihre Anwendung
Termine


30. – 31. März 2023
Padua, Italien


26. Mai 2023
München, Deutschland


21. – 24. Juni 2023
Zadar, Kroatien
Gründungsjahr
Ländern vertreten
installierte Systeme, weltweit
Jahre Erfahrung in TCD